Are You CovidMune?™
Pharmacological Inhibitors of the NLRP3 Inflammasome
Abstract
Inflammasomes play a crucial role in innate immunity by serving as signaling platforms
Introduction
In mammals, the immune system relies on innate immunity and adaptive immunity to protect the host from any external or internal danger (1). The innate immune response utilizes pattern-recognition receptors (PRRs) to sense endogenous or exogenous pathogens (2). A newly identified PRR, which was reported in detail for the first time in 2002, is the inflammasome. It is a high molecular weight protein complex
NLRP3 Inflammasome
NLRP3 inflammasome is the
NLRP3 Inflammasome recognizes a wide range of stimuli which include various protozoans, e.g., Plasmodium, ameba, viruses such as adenoviruses, influenza, and Sendai virus, fungi such as Saccharomyces cerevisiae and Candida albicans, different bacteria such as Listeria monocytogenes, Escherichia coli, and Staphylococcus aureus (14). NLRP3 Inflammasome can also respond to damage-associated endogenous factors such as drusen (15), uric acid crystals (16), extracellular adenosine triphosphate (ATP) (17), β-amyloid plaques (11), and islet amyloid polypeptide (18).
Activation of
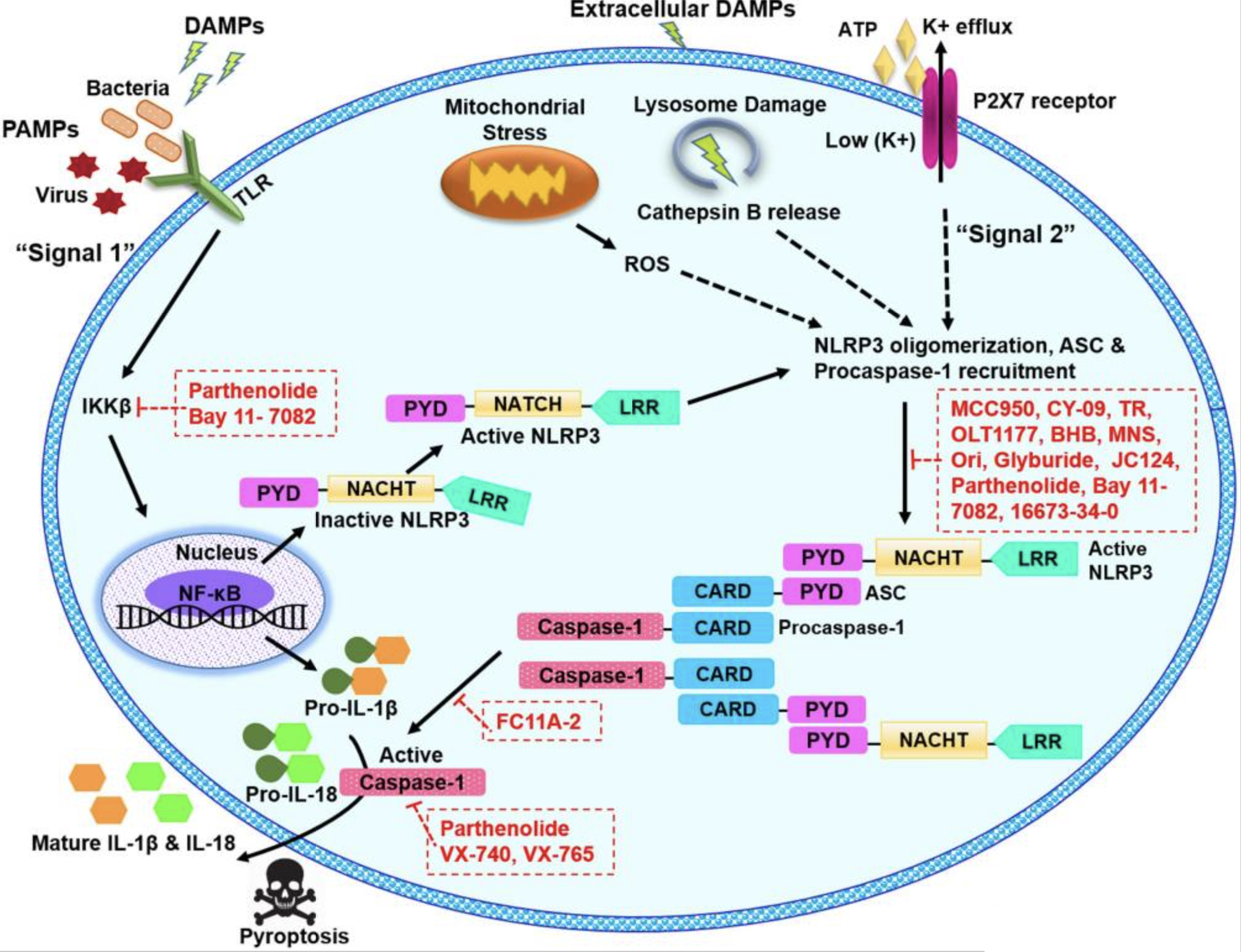
Figure 1
Schematic illustration of
As a result of the second step, caspase-1 is activated and carries out resultant processing and secretion of IL-1β and IL-18 (29). Various molecular mechanisms to explain the activation of NLRP3 inflammasome have been proposed which include mitochondrial reactive oxygen species (ROS) generation (30, 31), pore formation
NLRP3 Inflammasome Associated Diseases
Anomalous NLRP3 inflammasome activation is linked with the development of many diseases, especially age-associated ailments
Pharmacological Inhibition of NLRP3 Inflammasome
The association of NLRP3 inflammasome with the plethora of diseases evokes a substantial interest in the scientific community to discover the effective NLRP3 inflammasome inhibitors. By taking advantage of
Table 1
Potential inhibitors of NLRP3 inflammasome and their targets.
| Agent | Target(s) | Potential mechanism | References |
|---|---|---|---|
| Glyburide | NLRP3 (indirectly) | Inhibits ATP-sensitive K+ channels; downstream of P2X7 resulting in inhibition of ASC aggregation | (48, 54) |
| 16673-34-0 | NLRP3 (indirectly) | Induces NLRP3 conformational changes secondary to its activation or binding to ASC | (55, 56) |
| JC124 | NLRP3? | Blocks the expression of NLRP3, ASC, caspase-1, pro-IL-1β, TNFα and iNOS | (57) |
| FC11A-2 | NLRP3 (indirectly) | Interferes with proximity induced autocleavage of pro-caspase-1, suppresses IL-1β/18 release | (58) |
| Parthenolide | NLRP1, NLRP3 inflammasome, Caspase-1, NF-κB, IKKβ kinase activity | Alkylates cysteine residues in caspase-1 and in ATPase domain of |
(59, 60) |
| VX-740 | Caspase-1 | Covalent modification of the catalytic cysteine residue in the active site of caspase-1 resulting in caspase-1 blocking and resultant cleavage of pro-IL-1β/18 | (61, 62) |
| VX-765 | Caspase-1 | Covalent modification of the catalytic cysteine residue in the active site of caspase-1 resulting in caspase-1 blocking and resultant cleavage of pro-IL-1β/18 | (61, 63) |
| Bay 11-7082 | NLRP3, IKK, E2/3 enzymes, PTPs | Alkylates the cysteines in the ATPase domain of NLRP3, inhibits NLRP3 ATPase activity | (59, 64–66) |
| BHB | NLRP3 (Indirectly) | Inhibits K+ efflux resulting in reduced oligomerization of ASC and IL-1β/18 release | (49) |
| MCC950 | NLRP3 | Blocks the ATPase domain of NLRP3 resulting in inhibition of canonical and non-canonical NLRP3 inflammasome activation | (67, 68) |
| MNS | NLRP3 | Inhibits NLRP3 ATPase activity by cysteine modification, blocks NLRP3 inflammasome activation | (53) |
| CY-09 | NLRP3 | Inhibits NLRP3 ATPase activity, blocks NLRP3 inflammasome activation | (69) |
| Tranilast | NLRP3 | Binds to NLRP3 NACHT domain to block NLRP3-NLRP3 and NLRP3-ASC interaction | (52) |
| OLT1177 | NLRP3 | Inhibits NLRP3 ATPase activity, blocks NLRP3 inflammasome activation | (70) |
| Oridonin | NLRP3 | Binds to cysteine 279 of NLRP3 to abolish NLRP3-NEK7 interaction, blocks NLRP3 inflammasome activation | (71) |
Indirect Inhibitors
Glyburide
Glyburide is a sulfonylurea drug
16673-34-0
16673-34-0 is an intermediate substrate produced during glyburide synthesis, however, it lacks the
JC124
Kuwar et al. recently developed a novel small molecule JC124, through structural optimization of glyburide. JC124 was rationally designed to remove the potential hypoglycemic effects of glyburide. They explored the potential of JC124 for traumatic brain injury (TBI) therapy and it was demonstrated to exert significant anti-inflammatory effect to protect the injured brain following TBI. JC124 treatment significantly reduced the expression of NLRP3, ASC, caspase-1, pro-IL-1β, TNFα, and inducible nitric oxide synthase (iNOS). This targeting of NLRP3 inflammasome activation and its downstream neuroinflammatory cascade is suggested to confer JC124 its protective effect for TBI (57). It blocked ASC aggregation, caspase-1 activation, and IL-1β secretion. JC124 showed protective effects in a mouse model of acute myocardial infarction (77) and in transgenic AD models (77, 78). Further studies aiming at determining the efficacy of JC124 will render more information for its translational value.
FC11A-2
Liu et al. investigated a synthetic small molecular compound, 1-ethyl-5-methyl-2-phenyl-1H-benzo[d]imidazole, which is also known as FC11A-2, for its inhibitory potential of NLRP3 inflammasome. FC11A-2 was examined in THP-1 cells and in
Inhibitors for the Constituents of NLRP3 Inflammasome
Parthenolide
Parthenolide is a plant sesquiterpene lactone and has numerous anti-inflammatory properties, therefore, it is utilized in herbal medicines of various inflammatory diseases (79). It inhibited caspase-1 activation in response to NLRP1, NLRC4, and NLRP3 stimulation by alkylating many cysteine residues of caspase-1. Parthenolide can also target
VX-740 and VX-765
VX-740 (Pralnacasan) and its analog VX-765 are peptidomimetic inhibitor of caspase-1. They are prodrugs
Bay 11-7082
Bay 11-7082 is a phenyl vinyl sulfone, it inhibits
β-Hydroxybutyrate (BHB)
ß-
Direct Inhibitors of NLRP3 Protein
MCC950
A
MCC950 was reported to lower skin and pulmonary inflammation in mice (93) and some other in
3,4-Methylenedioxy-β-nitrostyrene (MNS)
A potent NLRP3 inhibitor, 3,4-Methylenedioxy-β-nitrostyrene (MNS) was found through screening a kinase inhibitory library by He et al. By utilizing immunoprecipitation, mass spectrometry, and mutational studies, it was demonstrated that MNS binds to the LRR and NACHT domains and suppresses ATPase activity of NLRP3, while the activation of AIM2 or NLRC4 inflammasomes was unaffected by it. MNS may directly target the cysteine(s) of NLRP3 as implicated by its inhibition of ATPase activity of NLRP3 (53). Future studies on MNS may confer additional insights on this potential inhibitor.
CY-09
Jiang et al. identified an effective and direct inhibitor of NLRP3 which showed significant inhibition of NLRP3 inflammasome in vivo in mice models and ex vivo in human cells (52). CY-09 is an analog of CFTR(
CY-09 demonstrated outstanding preventive or therapeutic properties in the mice models of gout, T2D, and CAPS. Most importantly, it exhibited a promising pharmacokinetic profile and showed good oral bioavailability, safety, and stability. Nonetheless, more studies are required to broaden its full potential (52).
Tranilast
Tranilast (N-[3′,4′-dimethoxycinnamoyl]-anthranilic acid, TR) is a tryptophan metabolite analog
OLT1177
OLT1177 is an active β-sulfonyl nitrile compound, which cleared phase I clinical trial for the treatment of degenerative arthritis successfully, and now being evaluated under phase II clinical trial (100).
A study in mice model of MSU- and zymosan-induced arthritis by Marchetti et al. demonstrated that OLT1177 has the potential to lower the neutrophil infiltration and joint swelling, as well as to inhibit the secretion of IL-1β and IL-6. In in vitro studies, OLT1177 blocked both canonical and non-canonical activation of NLRP3 inflammasome and showed direct binding with NLRP3 to block its ATPase activity. Moreover, in monocytes from CAPS patients, it lowered caspase-1 activity and resultant IL-1β
Oridonin
Oridonin (Ori) is a bioactive ent-
Concluding Remarks
NLRP3-induced pyroptosis and IL-1β/18 secretion
Author Contributions
AZ prepared the draft. All authors revised the draft.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Funding.
TJ was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (Grant No. XDB29030104), the National Natural Science Fund (Grant No. 31870731 and U1732109), the Fundamental Research Funds for the Central Universities, and the 100 Talents Programme of the Chinese Academy of Sciences. AZ was supported by
References
Articles from Frontiers in Immunology are provided here courtesy of Frontiers Media SA
